The next step was taken in England by William Huggins (1824-1910), a wealthy amateur astronomer who was a pioneer of astronomical spectroscopy. Comparing the spectrum of the comet 55P/Tempel-Tuttle with that of different products that he observed in the laboratory, he identified the bands discovered by Donati to those of carbon-containing molecules (in fact the dicarbon C2).
In 1881, obtaining more detailed spectra of another comet, he found the solar Fraunhofer lines, confirming Arago’s observation that the light of the comet is mainly scattered sunlight, but affected by emissions specific to the comet.

Dans ce spectre dessiné par Huggins en 1868 (en bas), on retrouve les bandes de Donati. Le spectre de la comète est comparé à des spectres de produits carbonés traversés par des étincelles. Les bandes sont dues à la molécule dicarbone C2.
Source : James LequeuxThe following years saw these ideas take shape slowly. Cometary spectra accumulated while laboratory work characterized the emissions of many new molecules. The identification of bands observed in cometary spectra progressed. Thus cyanogen CN was discovered in the great comet C/1881 K1; it was again observed in Halley’s comet in the following century.
The famous popularizer Camille Flammarion (1842-1925) disclosed the presence of this compound, causing a near panic. The night of 18-19 May 1910, when the Earth passed through the tail of the comet, people feared they would be victims of this violent poison. Flammarion denied the rumor, but it was too late! Of course, nothing happened.

Le premier spectre d’une comète obtenu par photographie dans le bleu et le violet, par Huggins en 1881. On y voit des bandes cométaires en émission (vers 40,5 et 42) et de nombreuses raies solaires en absorption qui montrent que la lumière cométaire est essentiellement de la lumière du Soleil diffusée.
Source : James LequeuxAfter much trial and error, scientists came to understand that the coma and tail of comets contain dust, and that it is this dust that scatters the sunlight. The dust grains are pushed by the radiation pressure exerted by the solar photons that strike them, a mechanism discovered by the great English physicist James Clerk Maxwell (1831-1879). The coma and tail also contain gas, where the following molecules were identified: C2, C3, CN and CH.
The next remaining question was: what’s inside the coma? This is what the Giotto probe discovered in 1986: a nucleus of ice and dust.
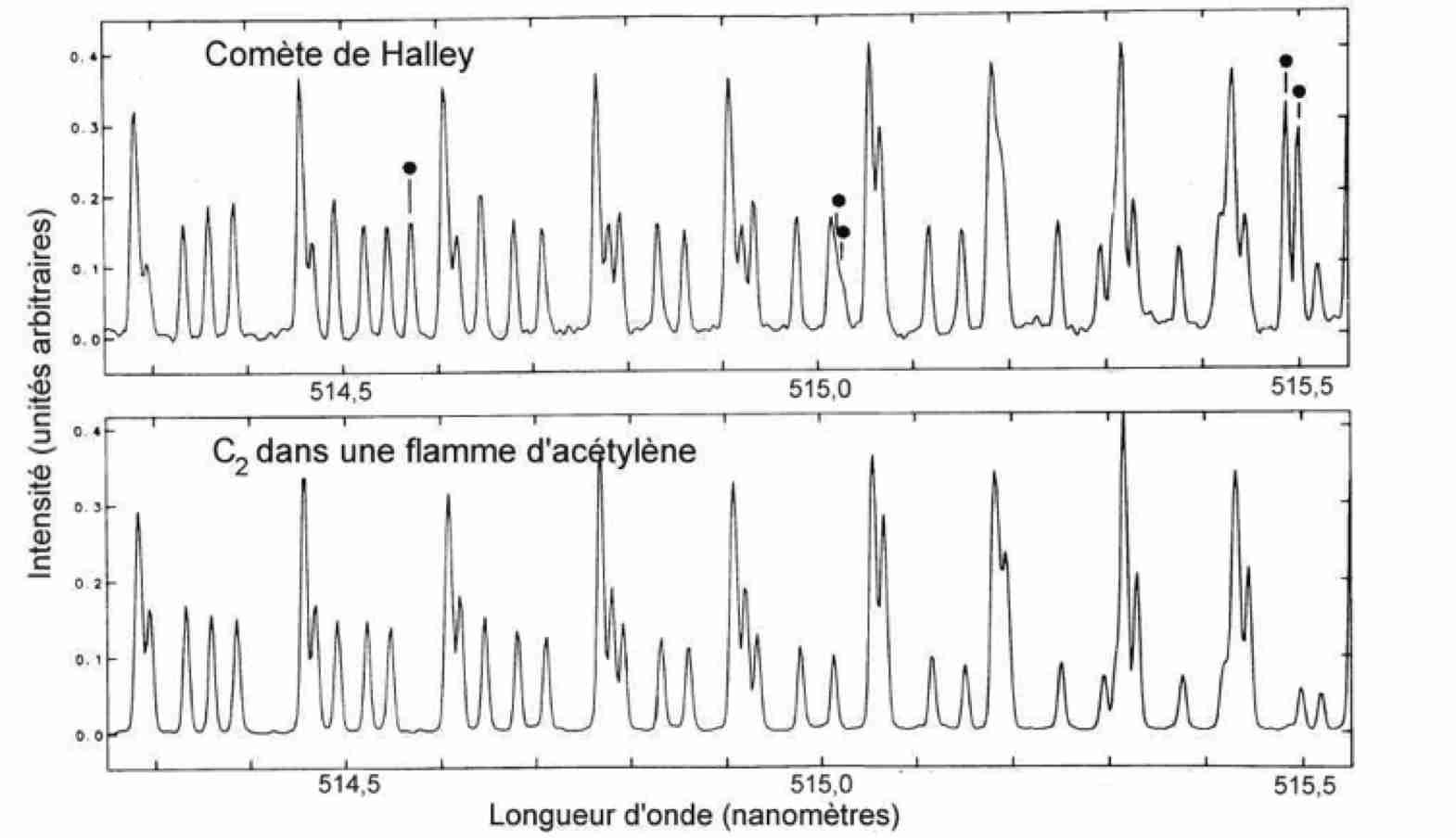
Un spectre à très haute résolution de la comète de Halley. Il est complètement dominé dans cet intervalle spectral par la bande d’émission de la molécule C2, ici décomposée en de très nombreuses raies. Quelques raies marquées d’un point proviennent de la molécule NH2. D’après D. Lambert et al. (1990), Astrophysical journal.
Source : James Lequeux